top of page

Methods and Resources
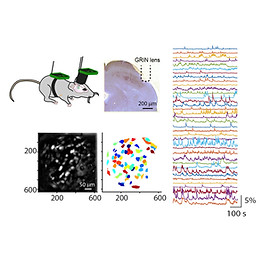
In the lab, we employ a broad range of techniques. Here, we outline some of the methods used in our work, as well as the resources we make available to the wider community. We run regular hands-on workshops so that everyone in the lab can pick up new skills as their projects evolve.
bottom of page